Porcelain versus Dental Enamel
The similarities and differences between dental porcelains and natural enamel.
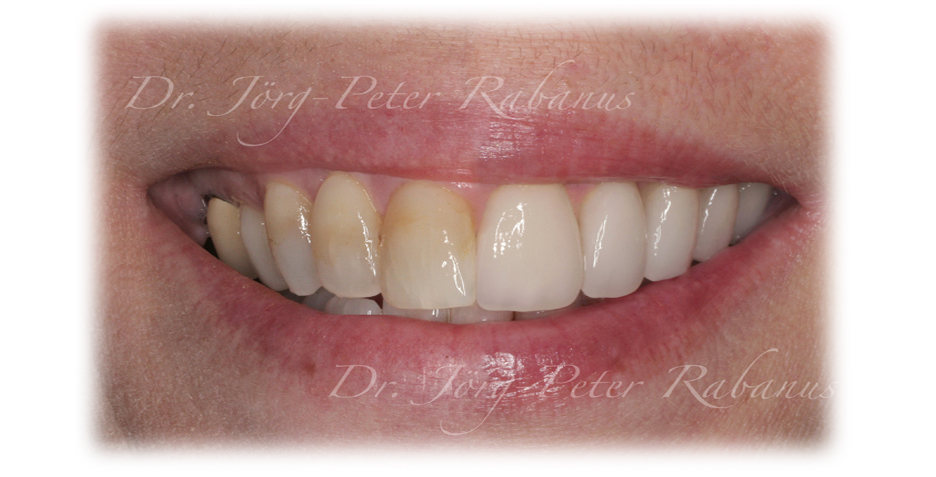
Tooth structure has been replaced with porcelains for well over two decades. Among those dental materials available, such as resin composites, alloys, and metal-ceramics, all-porcelain restorations are the most "toothlike" restorative materials. However, how closely do dental porcelains mimic the physical, biological, and optical properties of natural dental enamel?

The basic unit of enamel is the enamel rod. It has been well described in the book Oral Structure Biology (Embryology, Structure, and Function of Normal Hard and Soft Tissues of the Oral Cavity and Temporomandibular Joints) written by Hubert E. Schroeder for G. Thieme Verlag in 1991.
The enamel rod, or enamel prism, has a diameter of 4 to 8 micrometers. It is a tight agglomerate of organized hydroxyapatite crystals. It's cross section has a characteristic keyhole shape.
The enamel rod is built by ameloblasts, cells that are highly active during the formation of teeth. Enamel is a cell-free protective shield covering the coronal parts of mammalian teeth. It contains the fossilized footprint of the ameloblasts' activity throughout it's crystalline structure.
Ten Cate's Oral Histology, Elsevier, 2013, pages 122-128.
Deporter WA. The early mineralization of enamel. Calcified Tissue Research, 24 (1977) page 271.
Wakita M. The three-dimensional structure of Tomes' processes and the development of the microstructural organization of tooth enamel. In: Suga S. Mechanisms of Tooth Enamel Formation. Quintessence, Berlin, page 165.
Maas M. Built to Last: The Structure, Function, and Evolution of Primate Dental Enamel. Evolutionary Anthropology. Volume 8, Issue 4, pages 133–152, 1999
Warshawsky H. The development of enamel structure in rat incisors as compared to the teeth of monkey and man. Anat Rec 200 (1981) page 371.
Boyde A. The history of developing mammalian dental enamel. In Stack MV: Tooth Enamel (1965), pages 163,192.
Hoehling HJ. Die Bauelemente von Zahnschmelz und Dentin aus morphologischer, chemischer und struktureller Sicht. Hanser, Muenchen 1966.
For someone who understands oral biology and growth and development of the head and neck, tooth formation is a spectacle of nature. It is a firework of cascades of events that lead to a successive and highly organized mineralization of an organic matrix with hydroxyapatite micro-crystals. Cell-free technology will never be able to mimic nature and the orchestrated activity of various cell populations at this intricate level.

Observing the products of cell biology, it becomes apparent that living organisms are best equipped to create body parts whose physical, physiological, and mechanical properties match best the conditions of the environment. Genetics, epigenetics, intercellular communication, as well as the cells' perception of the environment lead to the most efficient solutions in response to environmental challenges.
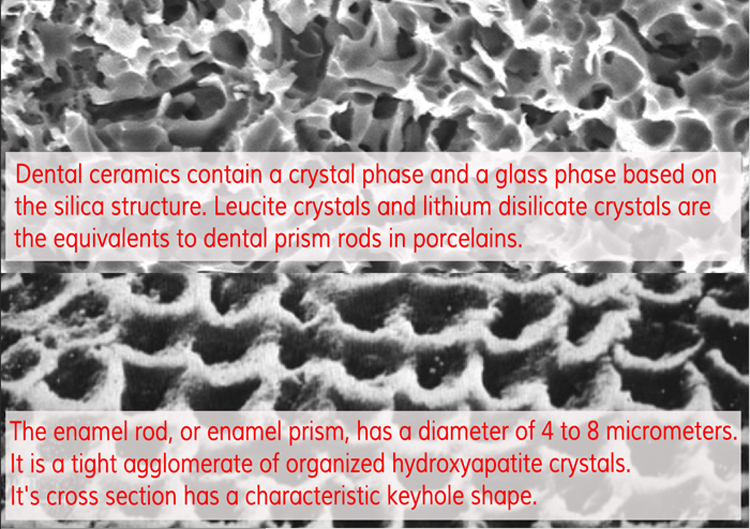
The equivalents to dental prism rods in porcelains are leucite crystals. Porcelain is created by vitrification of raw materials at high temperatures. Dental ceramics contain a crystal phase and a glass phase based on the silica structure. The thermal process known as ceraming consists of two steps:
1. Glass is heated up to 750 to 850ºC for one to six hours to form crystalline nuclei in the glass ("nucleation").
2. The temperature is increased to 1000 - 1150ºC for one to six hours, which is called the crystallizing point, which leads to the glazing of the porcelain.
Rosenblum MA. A review of all-ceramic restorations. J Am Dental Association 1997;128:297-307.
Anusavice KJ. Phillips Science of Dental Materials: Elsevier; 2004.
Adair PJ. Glass ceramic dental products. US Patent 4,431,420, 1984 - Google Patents.
A more recent glass ceramic contains lithium disilicate as the crystalline phase (Emax). It makes up about 70% of the volume of the porcelain. It has a unique microstructure where plate-like crystals interlock with each other and are randomly oriented. This leads to higher flexural strength via deflecting or blunting the propagation of cracks.
While porcelains are glasses that are not closely packed with micro-crystals as described here, enamel is a densely stacked conglomerate of hydroxyapatite crystals. The common dental ceramics are glassy in nature with short-range crystals. Hence, porcelains' molecular structure is more homogenous and less densely packed with micro-crystals than enamel, which coincides with a higher susceptibility to fracture. However, more recent developments in ceramic technology have yielded porcelains with a more randomized orientation of micro-crystals. Alumina porcelain is the only true crystalline ceramic and is one of the hardest and strongest oxides known. However, their aesthetic qualities are low.
The difference of molecular structure and assembly leads to differences in physical and optical properties. One major challenge for cosmetic dentistry is the attempt to mimic the natural metamerism of natural enamel, while it's physical properties are easier to be approximated.
Moormann S. Metamerism, morphogenesis, and the expression of Carabelli and other dental traits in humans. Am J Physical Anthropology, 2013 Mar;150(3):400-8.
Corcodel N. Metameric effect between natural teeth and the shade tabs of a shade guide. Eur J Oral Science, 2010 Jun;118(3):311-6.
When attempting to match the shade of a porcelain restoration to neighboring teeth, the color difference may not be the same under varying light conditions. This is called metamerism. The main reason why teeth are more opalescent than porcelains is the way how light get bounced around within the tooth. Porcelains might reflect light off it's surface just as enamel does at a certain angle. However, the difference of scattering of light within the body of a tooth and porcelains leads to differing light reflections at different light sources and angles of illumination. Hence, a careful three-dimensional mimicking of all dental layers with their individual optical characteristics will lead to the best approximation of natural opalescence in porcelain restorations. When designing dental veneers, the lab technician needs to understand where blue opalescent qualities appear within the body, how angled incisal edge facets scatter red-yellow wavelengths, how much light is kept within the body of porcelain of a certain optical density, and the fiber-optic effect of certain porcelains.
Rosenblum MA. A review of all-ceramic restorations. J American Dental Association, 1997;128:297-307.
Ceramics are classified by structure, by technique, and by composition. However, this would be a conversation among dentists . . . :)
Ceramics and glasses are brittle, which means that they display a high compressive and low tensile strength. The un-bonded porcelain may therefore be fractured at very low strains, such as 0.1 to 0.2%. It also has a much lower fracture toughness than other dental materials. Porcelains can be differentiated by the amount and types of crystals that have been added or grown in the glassy matrix. Common types are leucite, lithium disilicate or fluorapatite.
McLean. Evolution of dental ceramics in the twentieth century. J Prosthetic Dentistry, 2001 Jan; 85(1):61-6
Rizkalla AS. Indentation fracture toughness and dynamic elastic moduli for commercial feldspathic dental porcelain materials. Dental Materials, 2004 Feb; (20(2):198-206.
It should be emphasized at this point that porcelain veneers are only equivalent to natural enamel if they are intricately and expertly bonded to natural tooth structure. Bonding is a science and is dicussed on this page. As much as enamel is intricately attached to the underlying dentin, porcelain has the same requirements to display similar physical values. Here are some comparisons:

(1) Willems G. Composite resins in the 21st century. Quintessence Int. 1993 Sep;24(9):641-58.
(2) Giannini M. Ultimate tensile strength of tooth structures. Dental Materials, 2004 May;20(4):322-9.
(3) Johnston W. The shear strength of dental porcelain. J Dental Research, 1980 Aug;59(8):1409-11.
(4) Chung KH. The effects of repeated heat-pressing on properties of pressable glass-ceramics. J Oral Rehabilitation, 2009 Feb;36(2):132-41.

Lithium-disilicate filled glass ceramics. These filler particles are added mechanically during the starting glass by special nucleation techniques. They make up about 70% of the volume of the porcelain. It agglomerates as small interlocking plate-like crystals that are random oriented. Micro-fractures are deflected, branched, or blunted around the crystals, which is caused by a difference of thermal expansion between lithium-disilicate crystals and the glassy matrix, increasing tangential compressive stresses around the crystals. Hence, the propagation of micro-cracks is arrested. This property leads to a significant increase of flexural strength of the lithium-disilicate porcelain (350-450 MPa).
Deany IL. Recent advances in ceramics for dentistry. Crit Rev Oral Biology and Medicine, 1996; 7(2): 134-43.
Van Noort R. Introduction to dental materials. Philadelphia, PA: Elsevier Health Sciences; 2002. page 244.
Helvey GA. Retro-fitting an existing crown adjacent to a removable partial denture in a single visit. Inside Dentistry, 2009;5:34–41.
Mansour YF. Clinical performance of IPS-Empress 2 ceramic crowns inserted by general dental practitioners. J Contemporary Dental Practice 2008 May 1; 9(4):9-16.
Conrad HJ. Current ceramic materials and systems with clinical recommendations: a systematic review. J Prosthet Dent. 2007 Nov; 98(5):389-404.
Manicone PF. An overview of zirconia ceramics: basic properties and clinical applications. J Dent. 2007 Nov; 35(11):819-26.
Isabelle Denry. Ceramics for Dental Applications: A Review. Materials 2010, 3, 351-368.
Guazzato, M.; Albakry, M.; Ringer, S.P.; Swain, M.V. Strength, fracture toughness and microstructure of a selection of all-ceramic materials. Part I. Pressable and alumina glassinfiltrated ceramics. Dent. Mater. 2004, 20, 441–448.
Cattell MJ1J. The biaxial flexural strength of two pressable ceramic systems. Dent. 1999 Mar;27(3):183-96: Flexural strength of Empress 120 to 135 MPa.
Albakry M. Biaxial flexural strength, elastic moduli, and x-ray diffraction characterization of three pressable all-ceramic materials. J Prosthet Dent. 2003 Apr;89(4):374-80. Mean biaxial strengths were 175 +/- 32 MPa for Empress.
(To be continued . . .)



